-10% 🎉 for following me on Instagram! ![]()
New methods of immunohistochemistry: possibilities of modern diagnostics
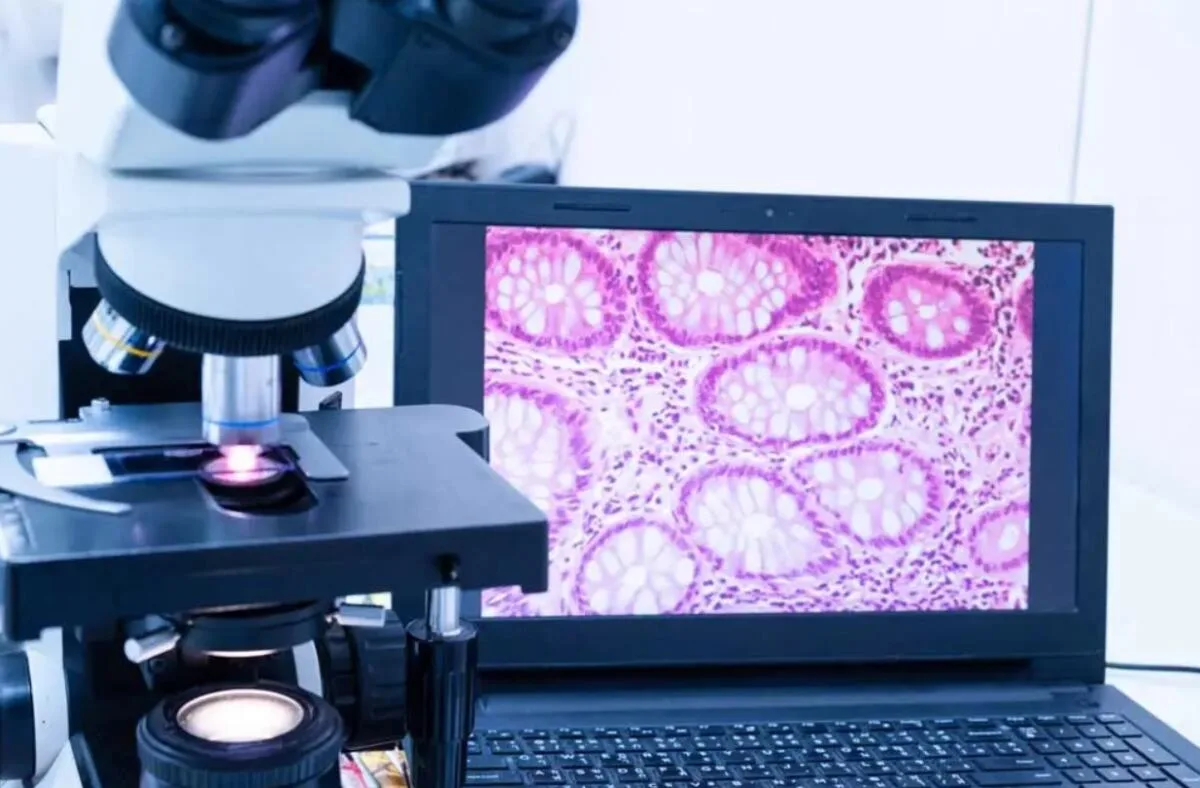
Morphological diagnostics of cancer and autoimmune diseases has reached a new level thanks to the development of immunohistochemistry (IHC). However, classical methods no longer always meet the requirements of accuracy, speed, and multiparametricity. Therefore, modern pathology is actively implementing new approaches that allow studying dozens of markers simultaneously, while preserving the context of the tissue.
New immunohistochemistry methods combine the advantages of classical histology, molecular biology, and digital technologies, creating opportunities for personalized medicine, particularly in oncology, neuroscience and immunopathology.
What is immunohistochemistry: a brief overview
Immunohistochemistry is a method that allows the detection of specific proteins (antigens) in tissue sections using labeled antibodies. The method is widely used for:
diagnosis of tumor type and subtype
prognosis of the disease
selection of targeted therapy
studying the expression of receptors, growth factors, tumor markers
Classical IGH is usually based on chromogenic reactions (e.g., DAB), but today the focus is on multiplex, digital, and automated technologies.
Latest technologies in immunohistochemistry
| Method | Essence | Advantages |
|---|---|---|
| Multiplex IHC (mIHC) | Simultaneous staining of multiple markers on one section | Sample saving, analysis of immune microenvironments |
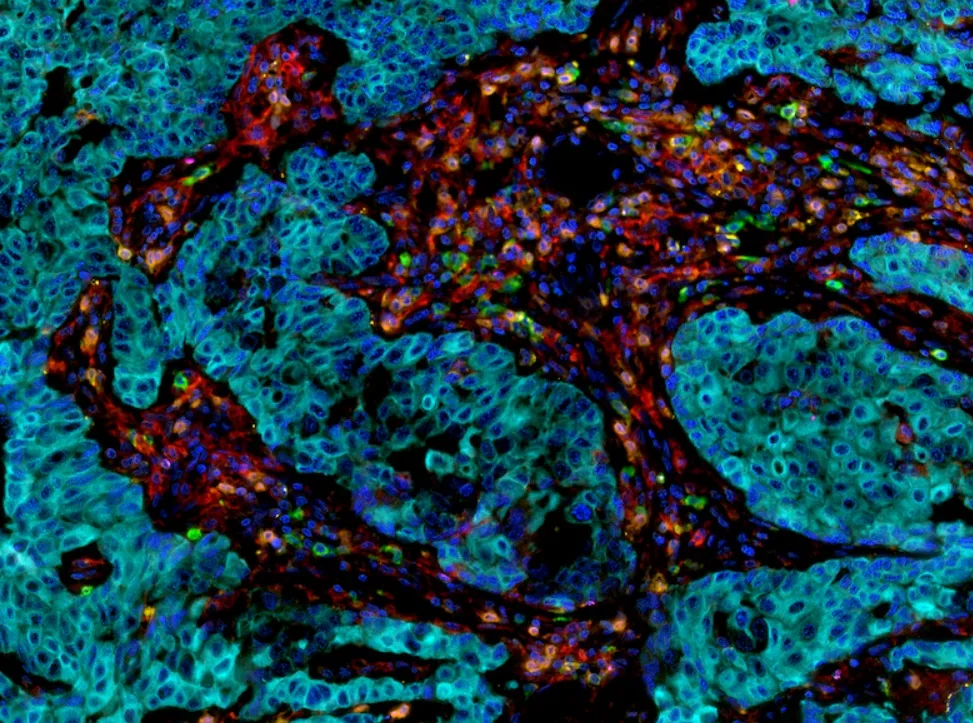
| Immunohistofluorescence (mIF) | The use of fluorescently labeled antibodies | High sensitivity, quantitative analysis |
| Tyramide Signal Amplification (TSA) | Signal amplification through enzymatic reaction | Extremely high sensitivity at low antigen levels |
| Digital Pathology + AI | Digitization of slices, automated analysis by algorithms | Minimization of subjective factors, high accuracy |
| GeoMx Digital Spatial Profiling | Combining IHC with mass spectrometry | Spatial profile of proteins and RNA in tissue |
| Nanostring IHC Panels | Analysis of dozens of markers in one cycle | Personalized oncopathology, immuno-oncology |
Application of the latest approaches
Oncology:
PD-L1: assessing the potential for immunotherapy
HER2, Ki-67: stratification in breast cancer
CD8/CD3/CD68: analysis of the tumor immune microenvironment
Neuropathology:
β-amyloid, phospho-tau: diagnosis of Alzheimer's disease
α-synuclein: for verification of Parkinson's disease
Hematology:
CD20, CD79a, CD5: classification of lymphomas
BCL2/BCL6/MYC: DLBCL differentiation and prognosis
Autoimmune diseases:
Cell segmentation, studying the phenotype of inflammatory infiltrates
Determining the ratio of T-regulators/effectors
Transplantology:
Identifying chronic rejection
Analysis of MHC expression and damage markers
Advantages of new methods
Reducing the use of biomaterial — important for small volume biopsies
Multivariate analysis — up to 40 markers on one slice
Automation — minimization of the human factor
Improving diagnostic accuracy — especially in difficult cases
Possibility of integration with molecular diagnostics (FISH, PCR, NGS)
Implementation difficulties
Despite the advantages, new technologies have their limitations:
High cost of equipment and reagents
The need for staff training
Lack of interpretation standards for new panels
Variability of results when preprocedural preparation is not followed
Limited availability in conventional laboratories
However, leading medical institutions are already implementing multiplex IGH as a routine, especially in oncology centers and neuropathology.
Modern immunohistochemistry techniques are opening up new depths in diagnostics: from the banal “positive/negative” to full-fledged tumor phenotyping, microenvironment analysis, and even prediction of response to treatment. Already today, they are forming the basis for truly personalized medicine — more accurate, faster, and more reliable than ever before.